|
6/13/2023 0 Comments Hertz cathode ray experiment He also showed that the nature of radio waves’ reflection and refraction was the same as those of light, and established beyond any doubt that light is a form of electromagnetic radiation obeying the Maxwell equations. In more advanced experiments, Hertz measured the velocity of electromagnetic radiation and found it to be the same as the light’s velocity. In this experiment Hertz confirmed Maxwell’s theories about the existence of electromagnetic radiation. The presence of oscillating charge in the receiver would be signaled by sparks across the (tiny) gap between the point and the sphere (typically, this gap was hundredths of a millimeter). This "receiver" was designed so that current oscillating back and forth in the wire would have a natural period close to that of the "transmitter" described above. He added a screw mechanism so that the point could be moved very close to the sphere in a controlled fashion. Hertz used a piece of copper wire, 1 mm thick, bent into a circle of a diameter of 7.5 cm, with a small brass sphere on one end, and the other end of the wire was pointed, with the point near the sphere. To prove there really was radiation emitted, it had to be detected. More information about this subject could be found in basic electronics text books. For an animated explanation of this device click here. This first radio waves transmitter is basically, what we call today, an LC oscillator. For his radio wave transmitter he used a high voltage induction coil, a condenser (capacitor, Leyden jar) and a spark gap - whose poles on either side are formed by spheres of 2 cm radius - to cause a spark discharge between the spark gap’s poles oscillating at a frequency determined by the values of the capacitor and the induction coil. The first clearly successful attempt was made by Heinrich Hertz in 1886. This challenged experimentalists to generate and detect electromagnetic radiation using some form of electrical apparatus. The most dramatic prediction of Maxwell's theory of electromagnetism, published in 1865, was the existence of electromagnetic waves moving at the speed of light, and the conclusion that light itself was just such a wave. Naturwissenschaftliche Rundschau, 12, 237–239.The Discovery of Electromagnetic RadiationĬonceptual Schematic of Hertz's Experiment Schriften der Physikalisch-Ökonomischen Gesellschaft zu Königsberg., 38, 3–20. Philosophical Magazine, Fifth Series, 48, 547–567. On the Masses of Ions and Gases at Low Pressures. Philosophical Magazine and Journal of Science, Fifth Series, 46, 528–45. On the Charge of Electricity Carried by the Ions Produced by Röntgen Rays. Proceedings of the Cambridge Philosophical Society, 9, 243–4. Philosophical Magazine and Journal of Science, Fifth Series, 44, 293–316. Philosophical Magazine and Journal of Science, Fifth Series, 38, 418–420. Of the “Electron” or Atom of Electricity. Scientific Transactions of the Royal Dublin Society, IV, 563–680. On the Cause of Double Lines and of Equidistant Satellites in the Spectra of Gases. Cambridge, MA: Dibner Institute for the History of Science and Technology. Warwick, Histories of the Electron: The Birth of Microphysics (pp. Revue générale des sciences pures et appliquées, 19, 386–402. Rendiconti del circolo matematico di Palermo, 21, 129–176. Comptes Rendus des Séances de l’Académie des Sciences, 121, 1130–34. Nouvelles propriétés des rayons cathodiques. CODATA Value: proton-electron mass ratio. National Institute of Standards and Technology. 
CODATA Value: electron charge to mass quotient. Proceedings of the Royal Society of London, 54, 438–461. 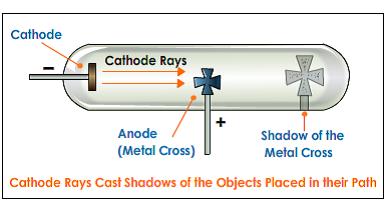
A Dynamical Theory of the Electric and Luminiferous Medium. Die magnetische Ablenkbarkeit der Kathodenstrahlen und ihre Abhängigkeit vom Entladungspotential. British Journal for the History of Science, 241–276. Thomson and the ‘Discovery of the Electron’. Corpuscles, Electrons and Cathode Rays: J. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
